|
ITER is backed by the EU, China, U.S., India, Russia, Japan and South Korea. This term, ITER says, refers to what happens when "the total power produced during a fusion plasma pulse surpasses the thermal power injected to heat the plasma." When it is up and running, those behind the project say it will generate net energy. ITER is centered around the development of a magnetic fusion device known as a tokamak.Īccording to the team at ITER, the tokomak "has been designed to prove the feasibility of fusion as a large-scale and carbon-free source of energy based on the same principle that powers our Sun and stars." "The larger French-based project and future power plants plan to use the same deuterium-tritium (D-T) fuel mix and operate under similar conditions to the record-breaking Eurofusion experiments held recently at Culham Science Centre, Oxford," they said. Those working on the JET project said the results reported this week were "a major boost" for the work being undertaken at the much bigger ITER project in southern France. Personal Loans for 670 Credit Score or Lower 3) Put them at the output, so you can dump the output ASAP. This works best when you are combining molecules / atoms into new molecules. This works best as a 'disassembly' reactor. Personal Loans for 580 Credit Score or Lower There are three philosophies on placements of the bond pads 1) Put them at the input, so you can disassemble the molecule quickly. Graphite has a layered, planar structure.Best Debt Consolidation Loans for Bad Credit Diamond and graphite, two forms of carbon and compounds like silicon dioxide and silicon carbide are all covalent networks. A covalent network structure consists of a giant 3-dimensional lattice of covalently bonded atoms.īoron, carbon and silicon are all examples of covalent network elements. Substances that consist of covalent molecules are usually gases or liquids at room temperature because the attractions between molecules are weak and easy to overcome.Ĭovalent substances that are solids with high melting points have much larger molecules. Oxygen molecules have a double bond: two shared pairs of electrons. Examples of these are diatomic oxygen (double bond) or nitrogen (triple bond). More than one bond can be formed between atoms leading to double and triple bonds. The oxygen forms two single covalent bonds with the two hydrogen atoms. Oxygen atoms have six outer electrons so need two more for a full outer shell. The shape formed is called trigonal pyramidal. :max_bytes(150000):strip_icc()/GettyImages-103311355-490ad3ba66d44d40b738a0e7d468ac8a.jpg) 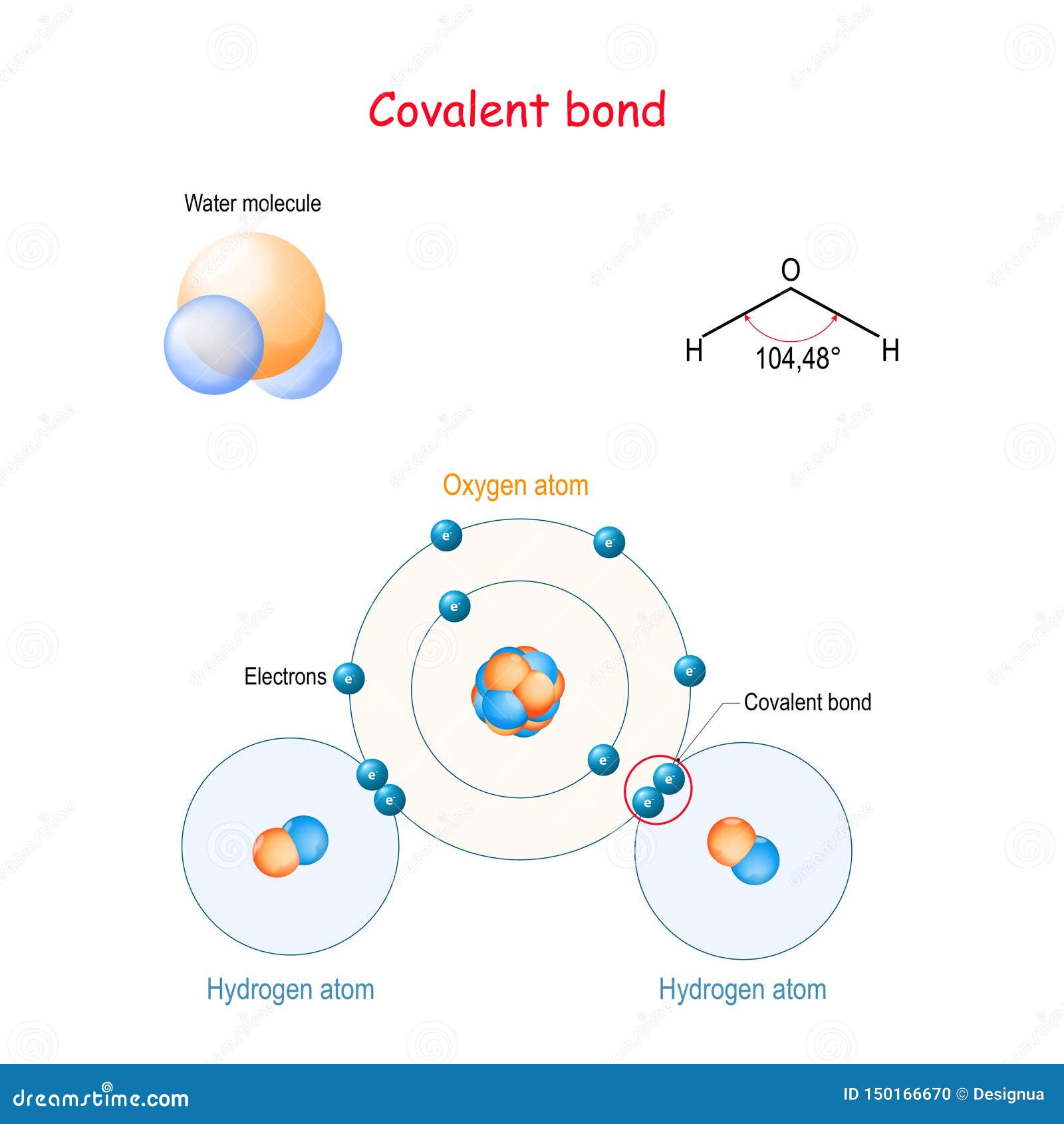
Nitrogen forms three single covalent bonds to hydrogen atoms. Nitrogen atoms have five outer electrons so needs three more for a full outer shell. A methane molecule has four shared pairs of electrons Ammonia (NH 3 ) The carbon forms four single bonds to the hydrogen atoms, so all the atoms now have a full outer shell of electrons. Methane (CH 4 )Ĭarbon atoms have four outer electrons so need four more for a full outer shell. A shared pair of electrons between two hydrogen atoms The shape of the molecule formed is called linear. Hydrogen (H 2 )īoth hydrogen atoms have only one electron, but by forming a single covalent bond, both can have a full outer shell. If you remember " I Br ing Cl ay F or O ur N ew H ouse” then you will have remembered that the seven diatomic elements are Iodine, Bromine, Chlorine, Fluorine, Oxygen, Nitrogen and Hydrogen.ĭiagrams can be used to show how the outer electrons are shared to form the covalent bonds in a molecule. There are seven diatomic elements that you have to remember and a simple mnemonic to help with this. A molecule is a group of atoms held together by covalent bonds.Ī diatomic molecule is a molecule containing only two atoms. Covalent bondingĪ covalent bond is a shared pair of electrons between atoms of two non-metal elements.Ī covalent bond happens when the positive nuclei from two different atoms are held together by their common attraction for the shared pair of electrons held between them.Ītoms that share pairs of electrons form molecules. There are different types of bonds that hold atoms together. To achieve a stable electron arrangement atoms can lose, gain or share electrons. 
When atoms form bonds, they can achieve a stable electron arrangement. Atoms can be held together by chemical bonds.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
